Brain Insulin Resistance & Weight Regain: Why Diets Fail

Your brain's insulin resistance is a leading reason why you can't stop regaining weight. If you've ever lost weight through meticulous calorie counting or macro tracking, only to watch the pounds creep back despite continued vigilance, you're not experiencing a failure of willpower. You're experiencing central nervous system insulin resistance.
This neurological phenomenon drives the relentless hunger, metabolic slowdown, and weight cycling that plague even the most dedicated dieters. Understanding it is crucial to breaking free from the regain cycle.
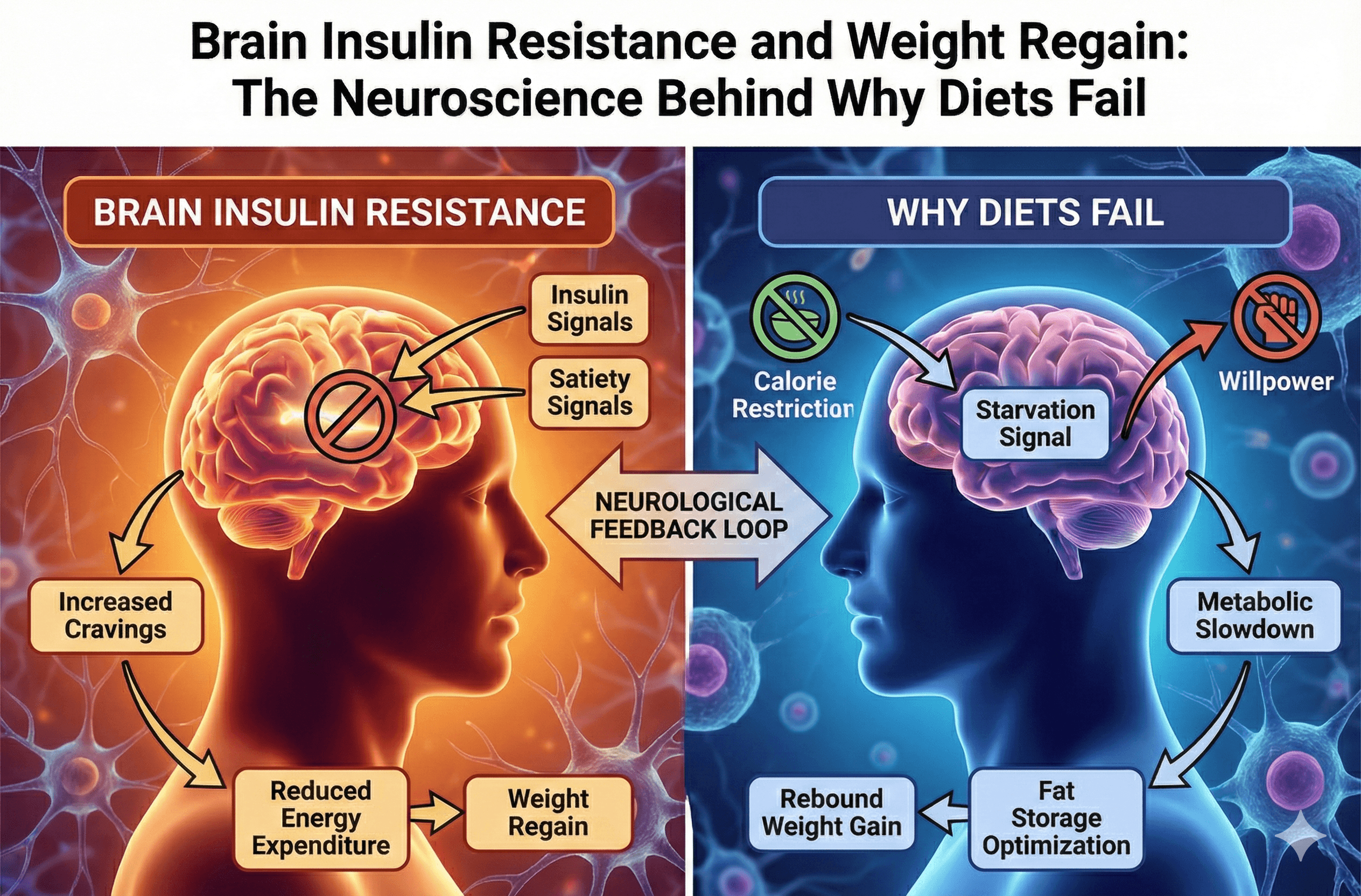
The standard narrative about weight regain usually focuses on peripheral insulin resistance, the well-known condition where your muscles, liver, and fat cells stop responding properly to insulin. However, emerging neuroscience reveals that insulin resistance in your brain, particularly in the hypothalamus, operates through entirely different mechanisms. It creates a powerful, biological drive to regain lost weight. This central insulin resistance doesn't just make weight loss harder; it actively sabotages your efforts by hijacking the very neural circuits that regulate hunger, satiety, and energy expenditure.
Act 1: Two Types of Insulin Resistance, Two Different Problems
To understand why brain insulin resistance is uniquely problematic for weight regain, we first need to distinguish it from the peripheral insulin resistance most people are familiar with.
Peripheral Insulin Resistance: The Metabolic Problem
When we talk about insulin resistance in conventional terms, we're usually referring to peripheral tissues, skeletal muscle, the liver, and adipose (fat) tissue. In these tissues, cells become less responsive to insulin's signal to absorb glucose from the bloodstream.
The pancreas compensates by producing more insulin, leading to chronically elevated insulin levels (hyperinsulinemia). You can learn more about how this affects the body through the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK). This is the insulin resistance associated with type 2 diabetes, metabolic syndrome, and cardiovascular disease.
Peripheral insulin resistance develops through well-characterized mechanisms:
- Chronic overnutrition: Particularly a high intake of refined carbohydrates and saturated fats.
- Cellular stress: Leading to inflammation and dysfunction in insulin receptor signaling pathways.
- The result: Impaired glucose disposal and severe fat storage dysregulation.
Central Insulin Resistance: The Neurological Problem
Brain insulin resistance, by contrast, occurs when neurons in the hypothalamus and other brain regions become unresponsive to insulin. This distinction is critical: your brain can be insulin resistant even while your peripheral tissues remain insulin sensitive, and vice versa.
The hypothalamus acts as your body's metabolic control center. It integrates signals about your energy status and orchestrates the appropriate responses. Normally, insulin in the brain serves as a satiety signal, it tells your hypothalamus that you have adequate energy stores and should reduce food intake. When hypothalamic neurons become insulin resistant, this vital satiety signaling fails.
Here is what makes brain insulin resistance particularly insidious: it can develop independently of peripheral insulin resistance and through entirely different mechanisms. While obesity and high-fat diets are risk factors for both, brain insulin resistance appears to be driven more strongly by:
- Hypothalamic inflammation: Saturated fats and overnutrition trigger inflammatory responses in the arcuate nucleus and other hypothalamic regions, which disrupts insulin receptor function.
- Endoplasmic reticulum (ER) stress: Neuronal cells experiencing metabolic stress show significantly impaired insulin signaling.
- Disrupted insulin transport: The blood-brain barrier's insulin transport mechanisms can become impaired, reducing the amount of insulin that actually reaches brain tissue.
- Reactive oxygen species: Oxidative stress in hypothalamic neurons interferes with insulin receptor substrate (IRS) proteins.
Crucially, brain insulin resistance can persist even after weight loss and improvement in peripheral insulin sensitivity. You can normalize your fasting glucose and HbA1c while your hypothalamus remains entirely insulin resistant, creating the perfect storm for weight regain.
Act 2: How Brain Insulin Resistance Drives Relentless Hunger
The connection between brain insulin resistance and weight regain centers on disrupted hunger signaling. It is a cascade of neurological dysfunction that makes sustained weight loss feel like fighting against your own biology (because you are).
The Insulin-Leptin Signaling Axis
To understand this mechanism, we need to examine how insulin and leptin work together in the hypothalamus. Leptin, often called the "satiety hormone," is produced by fat cells and signals to your brain about the size of your energy reserves.
In a healthy, properly functioning system:
Leptin from fat cells reaches the hypothalamus.
It activates POMC (pro-opiomelanocortin) neurons, which suppress your appetite.
It inhibits AgRP (agouti-related peptide) neurons, which stimulate hunger.
Insulin enhances leptin's effects, making the "I'm full" signal much stronger.
When your hypothalamus becomes insulin resistant, this entire system breaks down. Insulin can no longer amplify leptin's satiety signal, leading to profound downstream effects:
- Leptin resistance develops: Brain insulin resistance and leptin resistance are intimately linked. When hypothalamic neurons can't respond to insulin, they also become less responsive to leptin. Your fat cells are screaming, "We have plenty of energy!" but your hypothalamus can't hear the message.
- AgRP neurons become hyperactive: Without proper insulin signaling to suppress them, these hunger-promoting neurons fire excessively. This generates powerful hunger signals even when you have adequate or excess body fat.
- POMC neurons are suppressed: The appetite-suppressing neurons become less active, effectively removing a key brake on your food intake.
The Weight Regain Feedback Loop
This neurological breakdown creates a vicious cycle that explains why weight regain feels so inevitable:
You lose weight through caloric restriction: Your fat cells shrink and produce less leptin.
Leptin levels drop: This is a completely normal biological response to weight loss.
Your brain interprets this as starvation: Even if you're still carrying excess weight, the sudden drop in leptin signals an energy crisis.
Brain insulin resistance amplifies the response: Because your hypothalamus can't properly integrate insulin and leptin signals, it drastically overreacts to the leptin decrease.
Hunger increases dramatically: Your AgRP neurons go into overdrive.
Metabolic rate decreases: Your hypothalamus orchestrates a slowdown in energy expenditure (e.g., reduced thyroid output, decreased spontaneous movement, lowered body temperature).
The urge to eat becomes overwhelming: You experience this as obsessive food thoughts, intense cravings, and eventually, increased caloric intake.
Weight regain occurs: You gain back the lost weight, and very often, additional weight.
Brain insulin resistance worsens: The cycle of rapid weight gain and loss itself may further damage and worsen hypothalamic insulin resistance.
The Set Point Defense Mechanism
What we colloquially call your "set point", the weight your body seems determined to defend, is largely mediated by this exact hypothalamic insulin and leptin signaling system.
When brain insulin resistance is present, your defended set point may be elevated far above a healthy weight. Your hypothalamus essentially has a "miscalibrated thermostat" that believes you should weigh more than you currently do.
This is exactly why patients report that weight regain feels effortless while weight loss requires constant, exhausting vigilance. You're not imagining it. Your brain is actively working to restore your weight to its erroneously defended level through increased hunger and decreased energy expenditure.
Act 3: Restoring Brain Insulin Sensitivity—Breaking the Cycle
Understanding brain insulin resistance reframes weight regain from a personal failure to a neurological problem requiring targeted, clinical intervention. The crucial question becomes: How do we restore hypothalamic insulin sensitivity?
Dietary Approaches
Reduce Hypothalamic Inflammation Certain dietary patterns appear to actively reduce neuroinflammation in the hypothalamus:
- Limit saturated fat intake: High saturated fat diets are strongly linked to hypothalamic inflammation. Swapping them for monounsaturated fats (olive oil, avocados) and omega-3 fatty acids (fatty fish, algae) may cool down inflammatory signaling.
- Increase dietary fiber: Fiber and its fermentation products (like the short-chain fatty acid butyrate) have proven anti-inflammatory effects that may extend to the brain.
- Prioritize polyphenols: Compounds from berries, green tea, turmeric, and dark chocolate show promise in reducing oxidative stress in hypothalamic neurons.
Time-Restricted Eating Emerging research suggests that when you eat may matter as much as what you eat. Time-restricted feeding (consuming all calories within an 8-10 hour window) has been shown to improve hypothalamic insulin sensitivity in animal models. It may do this by:
- Reducing ER stress in hypothalamic neurons.
- Enhancing cellular cleanup mechanisms (autophagy).
- Synchronizing the circadian rhythms that regulate metabolic hormones.
Ketogenic Diets While sometimes controversial and difficult to sustain, ketogenic diets may improve brain insulin sensitivity by:
- Providing ketones (like beta-hydroxybutyrate), which have direct anti-inflammatory effects in the brain.
- Keeping carbohydrate intake very low, reducing the glycemic load that contributes to overall insulin resistance.
- Improving mitochondrial function in hypothalamic neurons through ketone metabolism.
(Note: The sustainability and long-term effects of keto require careful consideration and ideally, medical supervision.)
Exercise and Physical Activity
Exercise improves insulin sensitivity universally, but its effects on the brain are distinct from its effects on your muscles:
- Aerobic exercise: Increases blood flow to the brain and may improve how insulin is transported across the blood-brain barrier.
- High-intensity interval training (HIIT): Appears particularly effective at improving hypothalamic insulin sensitivity, potentially by increasing the production of brain-derived neurotrophic factor (BDNF).
- Resistance training: While it primarily improves peripheral insulin sensitivity, this indirectly benefits brain insulin signaling by reducing systemic inflammation.
The Golden Rule: Consistency is key. Intermittent, highly intense bursts of exercise are less effective than regular, moderate activity at maintaining long-term improvements in brain insulin sensitivity.
Sleep and Circadian Rhythm Optimization
Sleep deprivation is strongly associated with both peripheral and central insulin resistance. According to guidelines from the Centers for Disease Control and Prevention (CDC), poor sleep hygiene profoundly impacts overall metabolic health.
- Even a single night of poor sleep can impair hypothalamic insulin signaling.
- Chronic sleep restriction significantly increases hypothalamic inflammation.
- Circadian disruption (such as shift work or irregular sleep schedules) dysregulates the hormonal signals your hypothalamus relies on.
Practical Interventions:
- Prioritize 7 to 9 hours of quality sleep per night.
- Maintain consistent sleep and wake times, even on weekends.
- Minimize light exposure (especially screens and blue light) in the evening.
- Get morning bright light exposure to strengthen your circadian rhythms.
Stress Management
Chronic psychological stress activates inflammatory pathways and causes hypothalamic-pituitary-adrenal (HPA) axis dysfunction, which can severely worsen brain insulin resistance.
- Elevated cortisol: Directly interferes with insulin signaling in the hypothalamus.
- Stress-induced inflammation: Affects the exact hypothalamic regions involved in appetite regulation.
Evidence-Based Stress Reduction:
- Mindfulness meditation (proven to reduce systemic inflammation markers).
- Regular physical activity (offers a dual benefit for both insulin sensitivity and stress reduction).
- Building social connection and support systems.
- Scheduling adequate recovery and rest periods into your week.
Emerging Pharmacological Approaches
Lifestyle changes are foundational, but several medical compounds show incredible promise for directly targeting brain insulin resistance:
- GLP-1 Receptor Agonists (e.g., semaglutide, tirzepatide): These modern medications for type 2 diabetes and obesity appear to work partly through central nervous system mechanisms. They can potentially improve both hypothalamic insulin and leptin sensitivity, quieting the "food noise" in the brain.
- Intranasal Insulin: Delivers insulin directly to the brain, bypassing the blood-brain barrier. Clinical trials show improvements in appetite regulation, though long-term weight loss effects are still being studied.
- Metformin: While primarily targeting peripheral tissues, metformin may have beneficial secondary effects on brain insulin sensitivity and neuroinflammation.
These pharmacological options should always be considered under direct medical supervision as part of a comprehensive, multi-disciplinary approach.
The Long-Term Strategy
Restoring brain insulin sensitivity isn't a quick fix. Hypothalamic changes that develop over years of metabolic dysfunction simply won't reverse overnight. The most sustainable approach involves:
Gradual, modest weight loss: Aggressive caloric restriction may worsen brain insulin resistance through severe leptin drops and increased biological stress. Aim for a maximum of 0.5% to 1% of body weight lost per week.
Weight maintenance periods: Interspersing periods of weight loss with deliberate maintenance phases allows for hypothalamic adaptation and prevents severe leptin resistance.
Focus on metabolic health markers: Look for improvements in inflammatory markers, sleep quality, and hunger normalization. These often precede and predict sustainable weight loss.
Patience with the process: Hypothalamic remodeling may take 6 to 12 months (or longer) of consistent, daily intervention.
Maryland Trim Clinic (MTC) in Laurel, MD
Navigating the complexities of brain insulin resistance and weight regain is incredibly difficult to do alone. Having professional guidance ensures that you are treating the root neurological and metabolic causes of weight regain, rather than just treating the symptoms. At Maryland Trim Clinic, we understand that weight loss isn't a matter of willpower, but a complex medical journey.
Located in Laurel, MD, our clinic provides personalized, evidence-based care designed to reset your metabolic health safely. Whether you are looking for a comprehensive medical weight loss program to guide your nutrition and lifestyle habits, or you are a candidate for therapies like GLP-1 weight loss injections to help regulate your brain's hunger signaling, our team is here to support you. We focus on sustainable, long-term health improvements in a judgment-free environment. If you're tired of the endless cycle of yo-yo dieting, we invite you to explore how the team at our Maryland Trim Clinic homepage can help you build a strategy that works with your biology, not against it.
Conclusion: Reframing Weight Regain as a Neurological Issue
If you've struggled with weight regain despite doing "everything right," you are not weak-willed. You are experiencing the consequences of central nervous system insulin resistance. Your hypothalamus, the master regulator of energy balance, has become insensitive to the very signals that should tell it you have adequate energy stores.
This neurological dysfunction creates a powerful biological drive to regain weight through increased hunger and decreased metabolism. It's not simply about calories in versus calories out; it's about the neural circuits that determine what your brain perceives as the "right" number of calories and the "correct" body weight.
The path forward requires addressing brain insulin resistance directly through strategies that reduce hypothalamic inflammation, improve neural insulin signaling, and allow your metabolic control center to recalibrate. This means prioritizing dietary quality over sheer quantity, emphasizing consistency in sleep and exercise, managing stress, and considering medical interventions when appropriate.
Most importantly, it means abandoning the shame and self-blame that so often accompanies weight regain. You're not fighting a character flaw, you're addressing a complex neurometabolic condition. Understanding the neuroscience doesn't magically make the journey easy, but it does make it clear. And clarity is the first step toward sustainable change.
Disclaimer: The information provided in this article is for educational purposes only and is not intended as a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition or weight management plan.
Frequently Asked Questions
Q: Can I have brain insulin resistance without having type 2 diabetes or peripheral insulin resistance? A: Yes, absolutely. Brain insulin resistance can develop completely independently of peripheral insulin resistance. You can have normal fasting glucose, normal HbA1c, and good muscle insulin sensitivity while still having significant insulin resistance in your hypothalamus. This is one reason why some people with perfectly normal metabolic blood work still struggle with intense hunger and weight regain.
Q: How long does it take to reverse brain insulin resistance? A: The timeline varies significantly between individuals, but research suggests it takes considerably longer than reversing peripheral insulin resistance. While muscle insulin sensitivity can improve within days or weeks of dietary and exercise changes, hypothalamic insulin sensitivity may require 6 to 12 months or more of consistent intervention. This is because reversing structural changes in hypothalamic neurons, like inflammation and altered receptor function, takes time.
Q: Why do I feel hungrier after losing weight than I did before starting my diet? A: This is a hallmark of brain insulin resistance interacting with normal weight loss physiology. When you lose weight, leptin levels drop as your fat cells shrink. If your hypothalamus is insulin resistant, it can't properly integrate that remaining leptin signal with insulin signaling, leading to an exaggerated "starvation response." Your brain essentially misinterprets a modest drop in leptin as a severe energy crisis, triggering intense hunger that can feel far stronger than your pre-diet baseline.
Q: Are there any tests to diagnose brain insulin resistance? A: Currently, there is no simple clinical blood test for brain insulin resistance available for routine use. It is typically inferred from clinical presentation: unexplained weight regain despite dietary adherence, intense hunger that seems disproportionate to caloric intake, a history of weight cycling, and sometimes resistance to traditional weight loss medications. Your symptoms and pattern of weight regain remain the best clinical indicators.
Q: Will intermittent fasting help with brain insulin resistance? A: Time-restricted eating (a form of intermittent fasting) shows excellent promise for improving brain insulin sensitivity. Animal studies demonstrate that restricting eating to specific time windows reduces hypothalamic inflammation while improving insulin signaling in the brain. The mechanism appears related to enhanced cellular cleanup (autophagy) and better circadian rhythm alignment. If you try it, start with a 12-hour eating window and gradually narrow it to 8-10 hours, ensuring you don't trigger excessive hunger.
Ready to Break the Cycle of Weight Regain?
If you are tired of fighting your biology and want a sustainable, medically supervised approach to metabolic health, we can help. Schedule a consultation with the experts at Maryland Trim Clinic today to discuss personalized strategies, advanced testing, and medical weight loss solutions designed for your unique physiology.