Zepbound vs. Retatrutide Side Effects: Trade-offs Explained

Retatrutide is being called the "Godzilla" of weight loss medications, but does more power mean worse side effects than Zepbound?
If you are already utilizing tirzepatide (Zepbound) and successfully managing your weight or metabolic health, you have probably heard the buzz. A new drug is on the horizon that reportedly outperforms every GLP-1 medication currently on the market.
Retatrutide, developed by Eli Lilly, generated enormous excitement in the medical community after Phase 2 trial results showed jaw-dropping weight loss numbers. But for patients who have already wrestled with nausea, fatigue, and GI distress on Zepbound, the real question isn't just how much weight can I lose?—it's what will my body have to go through to get there?
This article breaks down the mechanistic differences between these two drugs, digs into the actual side effect data from clinical trials, and helps you figure out whether retatrutide represents a meaningful upgrade or a heavier burden for your body.
ACT 1: The Triple Threat — Understanding Why Retatrutide Is Built Differently
Zepbound's Dual Mechanism: A Quick Recap
Zepbound (tirzepatide) works as a dual agonist, meaning it activates two hormonal receptors simultaneously:
- GLP-1 (glucagon-like peptide-1): Slows gastric emptying, reduces appetite, and improves insulin sensitivity.
- GIP (glucose-dependent insulinotropic polypeptide): Enhances insulin secretion, alters how the body stores fat, and appears to soften the tolerability of harsh GLP-1 side effects.
This dual mechanism is precisely why GLP-1 weight loss injections like Zepbound consistently outperformed semaglutide (Wegovy) in head-to-head trials. It doesn't just add efficacy; it balances the bodily response.
Retatrutide's Triple Mechanism: Adding a Third Engine
Retatrutide goes one step further by adding a third receptor target. It is a triple agonist:
GLP-1 receptor agonism
GIP receptor agonism
Glucagon receptor agonism (the new ingredient)
Glucagon is typically thought of as the hormone that raises blood sugar, acting in opposition to insulin. So why activate it in an appetite suppressant medication? The answer lies in energy expenditure.
Glucagon receptor activation significantly increases your metabolic rate, promotes fat breakdown in the liver, and enhances thermogenesis (burning energy as heat). Retatrutide doesn't just tell your brain you are less hungry; it simultaneously tells your liver and fat tissue to burn more aggressively.
But activating a third hormonal pathway introduces a third category of potential physiological disruption—and that is the trade-off.
ACT 2: The Data Deep Dive — What Phase 2 Trials Actually Show
The Efficacy Numbers
The Phase 2 trial of retatrutide published in the New England Journal of Medicine (2023) made headlines for good reason. Over 48 weeks, participants at the highest dose lost an average of 24.2% of their body weight. For context, Zepbound's Phase 3 trial showed approximately 20.9% weight loss over 72 weeks. Retatrutide achieved superior results in a much shorter timeframe.
But weight loss percentages do not tell you what it actually felt like to be a participant.
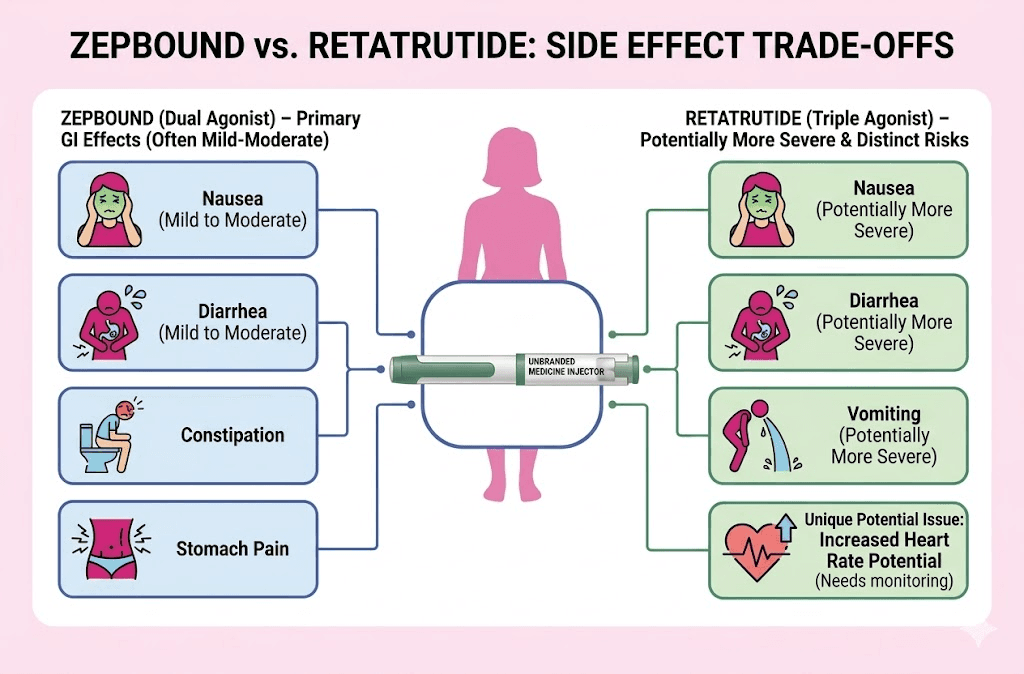
Gastrointestinal Side Effects: The Most Important Comparison
GI side effects remain the primary reason patients discontinue weight loss therapies. Here is how the two drugs compare based on clinical data at their highest respective doses:
Retatrutide Phase 2 (12 mg dose):
- Nausea: ~42% of participants
- Vomiting: ~25%
- Diarrhea: ~22%
- Constipation: ~20%
- Discontinuation due to GI events: ~16%
Zepbound Phase 3 (15 mg dose):
- Nausea: ~31–33%
- Vomiting: ~18–20%
- Diarrhea: ~20–22%
- Constipation: ~17–18%
- Discontinuation due to GI events: ~6.9%
The Reality Check: The pattern is clear. Retatrutide produces meaningfully higher rates of GI side effects (particularly nausea and vomiting) and significantly higher discontinuation rates. This is consistent with what pharmacologists would predict: more receptor activation equals more physiological perturbation.
The Glucagon Component: Unique Side Effects
The glucagon receptor agonism in retatrutide introduces side effects not typically seen with Zepbound:
- Heart rate elevation: Retatrutide produced more pronounced resting heart rate increases (5–10 bpm above baseline in some users). This is a critical watchpoint for patients with cardiac rhythm concerns.
- Liver enzyme fluctuations: Because glucagon acts directly on hepatic tissue, some patients showed transient increases in liver enzymes during early dose escalation.
- Blood glucose variability: Glucagon's glucose-raising effects can create mild fluctuations that some people feel as shakiness or low-grade anxiety.
Where Retatrutide May Actually Be Gentler
It's not a completely one-sided story. Preliminary data suggests that retatrutide's glucagon component may actually improve energy levels and reduce the severe fatigue that patients on pure GLP-1 therapies often report. By mobilizing fat for energy, it helps bridge the caloric deficit gap. Additionally, early data suggests retatrutide shows remarkable promise in addressing fatty liver disease (NASH).
ACT 3: The Verdict — Should You Stay on Zepbound or Wait for Retatrutide?
Stick With Zepbound If:
You've achieved meaningful results with manageable side effects. If you're losing weight steadily and your nausea has resolved, there is no compelling reason to switch to a drug with a higher documented GI burden.
You have a history of GI sensitivity. Patients who struggled significantly during Zepbound titration should approach retatrutide with extreme caution.
You have cardiac rhythm concerns. The heart rate elevations associated with retatrutide make it a risky choice for anyone with a history of arrhythmia or tachycardia.
You want a mature safety profile. Zepbound has gone through massive Phase 3 trials and years of real-world use.
Consider Retatrutide If (Once Approved):
You have plateaued on maximum-dose Zepbound. If weight loss has stalled significantly short of your medical goals, retatrutide's additional metabolic firepower may be necessary.
You have severe fatty liver disease. The hepatic benefits of glucagon receptor agonism appear to give retatrutide a meaningful advantage here.
You sailed through Zepbound's side effects. Patients who experienced zero or minimal nausea on Zepbound are best positioned to tolerate retatrutide's higher GI burden.
(Note: Because of the aggressive nature of triple-agonist weight loss, patients must be hyper-vigilant about protecting lean mass. Incorporating muscle building and toning routines is non-negotiable to prevent severe muscle degradation).
Maryland Trim Clinic (MTC) in Laurel, MD
The rapid evolution of weight loss medications—from GLP-1 monotherapies to dual and triple agonists—means that choosing the right prescription is more medically complex than ever. Located in Laurel, MD, the Maryland Trim Clinic (MTC) provides the comprehensive clinical oversight required to match your unique physiology with the appropriate medication.
At MTC, we don't just write prescriptions; we actively manage your side effect profile and long-term health trajectory. When you enroll in a tailored medical weight loss program, our clinical team utilizes advanced tools like metabolic testing and analysis to monitor your heart rate, liver function, and body composition. Combined with dedicated nutritional counseling and coaching to mitigate severe GI distress, MTC ensures that your weight loss journey is safe, effective, and perfectly calibrated to your body’s tolerance.
The Bigger Picture: What 'Godzilla' Actually Means
The "Godzilla" nickname captures something real about retatrutide—it is a larger, more powerful intervention with a proportionally larger footprint on your physiology. That isn't inherently bad. For some patients, maximum firepower is exactly what their health situation demands.
However, power without precision carries costs. The U.S. Food and Drug Administration (FDA) will require extensive Phase 3 data before approving retatrutide, ensuring its risk-benefit profile is fully understood.
Neither drug is universally superior. Zepbound remains a remarkably effective, well-tolerated medication. Retatrutide represents a genuine next-generation advance that will likely serve a different, more metabolically resistant patient population. The era of personalized metabolic medicine is here, and understanding why these drugs differ is the first step toward making the right choice for your body.
Medical Disclaimer: Retatrutide has not received FDA approval as of the time of publication; all clinical data referenced is from Phase 2 trials. The information provided in this article is for educational purposes only and is not intended as a substitute for professional medical advice. Always consult a qualified healthcare provider before making changes to your medication regimen.
Frequently Asked Questions
Q: What is retatrutide and how is it different from Zepbound? A: Retatrutide is an investigational triple-agonist drug that targets GLP-1, GIP, and glucagon receptors simultaneously. Zepbound (tirzepatide) is a dual-agonist that targets only GLP-1 and GIP. The addition of glucagon increases fat burning but also broadens the potential side effects.
Q: Does retatrutide cause more nausea than Zepbound? A: Based on Phase 2 clinical data, yes. Retatrutide produced nausea in approximately 42% of participants at the highest dose, compared to roughly 31–33% in Zepbound's highest dose Phase 3 trials. Vomiting and discontinuation rates were also notably higher.
Q: Is retatrutide FDA-approved yet? A: No. As of early 2026, retatrutide remains an investigational drug moving through Phase 3 clinical trials and has not yet received FDA approval for public use. Zepbound is fully FDA-approved.
Q: Why does retatrutide cause heart rate increases? A: Glucagon naturally stimulates the heart as part of its metabolic "fight-or-flight" role. Retatrutide produced more pronounced resting heart rate increases (5–10 bpm) than Zepbound, making it a critical consideration for patients with cardiac conditions.
Q: Can retatrutide help with fatty liver disease in a way Zepbound cannot? A: Early data suggests yes. The glucagon receptor component promotes fat breakdown directly in liver tissue, showing promising signals for improving non-alcoholic steatohepatitis (NASH) more aggressively than dual-agonists.
Q: Will retatrutide replace Zepbound when it's approved? A: Unlikely. They will serve different populations. Zepbound offers strong efficacy with a moderate side-effect burden. Retatrutide will likely be reserved for patients who plateau on Zepbound, require maximum weight loss, or have specific conditions like NASH.
Ready to Optimize Your Weight Loss Strategy?
Don't guess which medication is right for your body. Visit the Maryland Trim Clinic homepage today to schedule a comprehensive clinical evaluation. Our expert medical team in Laurel, MD, will help you understand your options and build a safe, sustainable path to your health goals.